 
Revision notes and formula sheets are shared with you, for grasping the toughest concepts. WAVE platform encourages your Online engagement with the Master Teachers. We provide you year-long structured coaching classes for CBSE and ICSE Board & JEE and NEET entrance exam preparation at affordable tuition fees, with an exclusive session for clearing doubts, ensuring that neither you nor the topics remain unattended. We have grown leaps and bounds to be the best Online Tuition Website in India with immensely talented Vedantu Master Teachers, from the most reputed institutions. 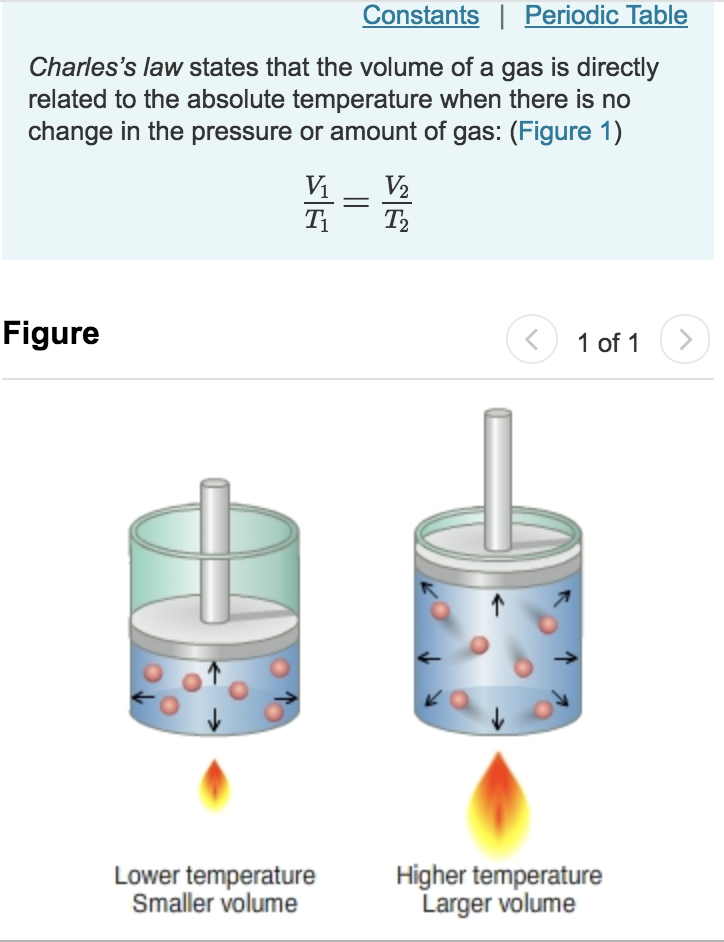
Vedantu LIVE Online Master Classes is an incredibly personalized tutoring platform for you, while you are staying at your home. To answer questions related to Charles law, you need to focus on clarifying your doubts. Gas laws are defined at idea conditions and are then utilized in real-time situations for making proper calculations. When the temperature decreases, the volume will decrease, and vice versa.Īccording to the above statement, we can write the following when comparing a substance in two different circumstances: His unpublished work provides extensive details about this law.Ī detailed explanation of how gas expands when the temperature rises is provided by Charles' Law, also known as the law of volumes. According to the law, when constant pressure is applied to a sample of dry gas, the Kelvin temperature, and the volume will be directly proportional.Ī French physicist named Jacques Charles first proposed this law in the year 1780. How do you express Charles Law in math?Ĭharles’s law states that the volume of a gas is inversely proportional to the absolute temperature.As the temperature and pressure of an ideal gas remain constant, Charles' law states that the volume is directly proportional to its absolute temperature at constant pressure. You can also use a manometer, which is a device that measures the difference in pressure between two points in a gas. This is a thermometer that measures the pressure of a gas. To prove Charles’ Law, you can use a gas thermometer. The law is a result of the kinetic theory of gases, which states that gas particles are in constant motion. This law is named after Jacques Charles, who discovered it in 1787. How do you prove Charles Law?Ĭharles’ Law is a scientific law that states that when a gas is heated, its pressure will increase. This results in the gas taking up more space. Conversely, when the temperature of a gas is decreased, the molecules move slower and collide with each other less frequently. This results in the gas taking up less space. When the temperature of a gas is increased, the molecules move faster and collide with each other more frequently. This law is due to the fact that the molecules of a gas are in constant motion. 
Conversely, when the temperature of a gas is decreased, the volume of the gas increases. This means that when the temperature of a gas is increased, the volume of the gas decreases. Read also Insurrection Act Vs Martial Law 2021 What happens to the molecules in Charles Law?Ĭharles Law states that the volume of a gas is inversely proportional to the absolute temperature of the gas. For example, if you know that a gas is being heated, you can use the equation to predict how the gas will change in volume. The Charles’ Law equation can also be used to predict the behavior of gases under different conditions. Conversely, if you know the temperature and pressure of a gas, you can use the equation to calculate the gas’s volume. For example, if you know the temperature and volume of a gas, you can use the equation to calculate the gas’s temperature. The Charles’ Law equation can be used to solve problems involving gases. This law is usually represented by the equation V=kT, where V is the volume of the gas, k is a constant, and T is the temperature. How do you use Charles Law?Ĭharles’ Law is a gas law that states that, at a constant pressure, the volume of a gas is inversely proportional to its temperature. As the air in the balloon heats up, the balloon will get smaller, since the volume of the gas inside the balloon will decrease. Suppose you have a balloon that is filled with air. 
The following example illustrates Charles’ law. This law is named after French physicist Jacques Charles, who first published it in 1787. In other words, as the temperature of a gas increases, its volume decreases. What is Charles Law explain with example?Ĭharles’ law is a physical law that states that the volume of a gas is inversely proportional to the absolute temperature of the gas. The Charles Law is one of the basic principles of thermodynamics, and it helps us to understand how heat is transferred between gases and other systems. Thermodynamics is the study of the energy and heat transfer between systems. The Charles Law is also important in the field of thermodynamics. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
